NMM/OMT
BRIEF REPORT
Augmentation of immune response to vaccinations through osteopathic manipulative treatment: a study of procedure
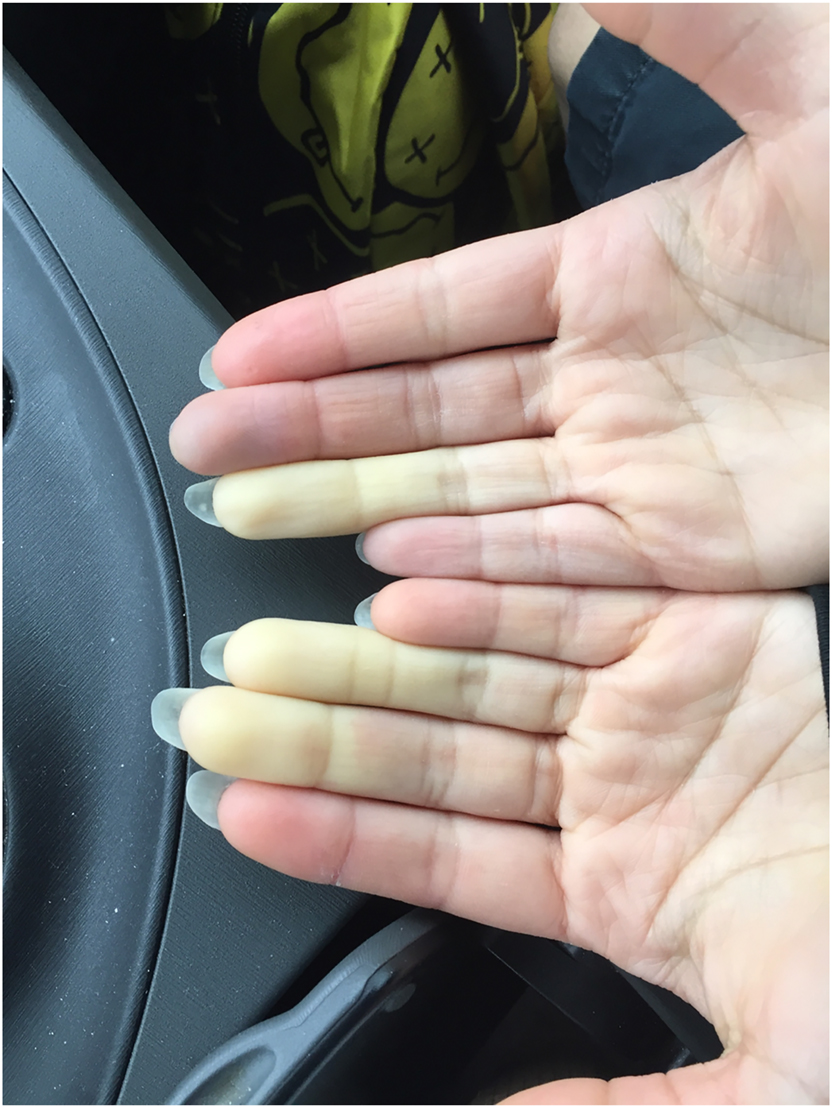
This article describes a proposed protocol to evaluate the effects of osteopathic manipulative treatment on antibody generation in the peripheral circulation in response to a vaccine and its possible …